Patents
- Appayee, C.; Padmaja, V. M. D.; Kutwal, M. S. “Photochromic organic compounds and a process for preparation thereof”. Indian Patent, Application No. 202021012831.
- Appayee, C.; Padmaja, V. M. D. “Photochromic organic compounds and a process for preparation thereof”. Indian Patent, Application No. 202021012829, dated March 24, 2020, issued as Patent No. 498888, dated January 12, 2024.
- Appayee, C.; Sarkale, A. M. “Method for the asymmetric synthesis of (S)-paraconic acid”. Indian Pat. Appl. No. 201721019540, dated June 3, 2017, issued as Patent No. 391941, dated March 14, 2022.
Publications from IITGN
Singh, S.; Kumar, R.; Dubey, N.; Appayee, C.* Synthesis and Application of Chiral cis-2,5-Disubstituted Pyrrolidine Organocatalysts” Chem. Commun. 2024, Article ASAP. Read more…
Invited Article for the themed collection “2024 Pioneering Investigators” Read more…

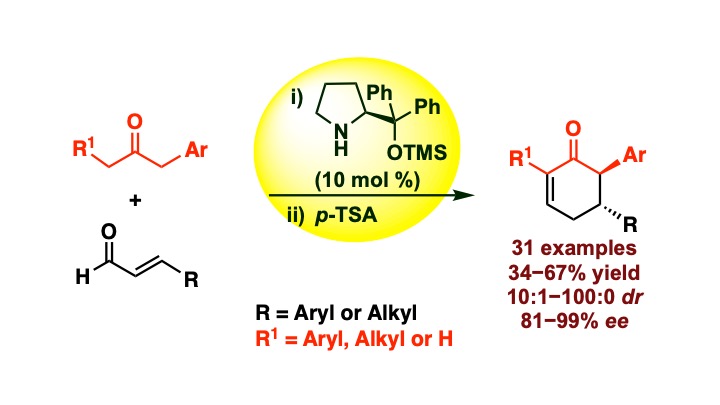
Mehta, R.; Kumar, R.; Singh, S.; Appayee, C.* “Asymmetric Synthesis of α-Arylcyclohexenones Catalyzed by Diphenylprolinol Silyl Ether” J. Org. Chem. 2024, Article ASAP. Read more…
Kumar, R.; Maurya, V.; Avinash, A.; Appayee, C.* “Nonsilyl Bicyclic Secondary Amine Catalysts for the Asymmetric Transfer Hydrogenation of α,β-Unsaturated Aldehydes” J. Org. Chem. 2024, 89, 8586–8600. Read more…

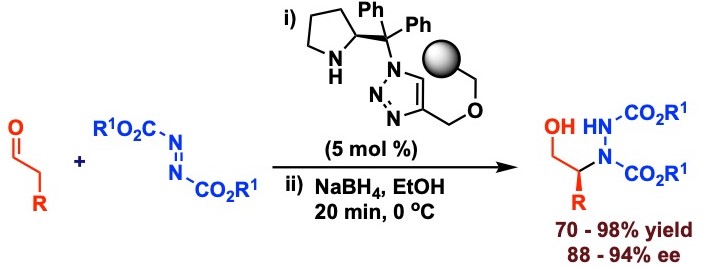
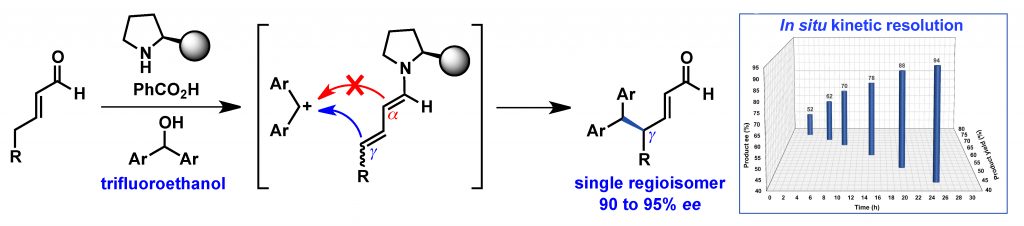
Kumar, R.; Singh, S.; Mehta, R.; Appayee, C.* “Polystyrene-Supported Aminocatalyst Derived from Diarylprolinol for Asymmetric α-Amination of Aldehydes” Eur. J. Org. Chem. 2023, 26, e202300147. Read more…
Invited Article for a special collection “Asymmetric Synthesis and Catalysis” Read more…
This article has been highlighted on the Front cover page of the journal. Read More…
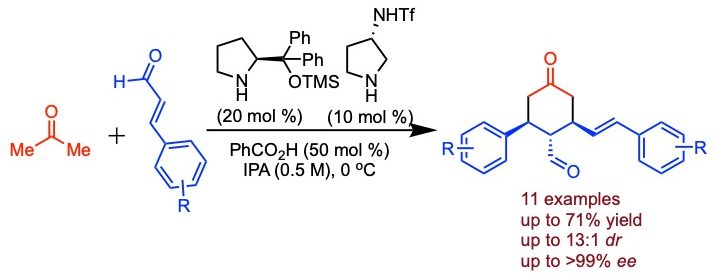
Maurya, V.; Kutwal, M. S.; Appayee, C.* Direct Catalytic Asymmetric Synthesis of Disubstituted 4-Oxocyclohexanecarbaldehydes from Acetone and Cinnamaldehyde Derivatives” Org. Lett. 2021, 23, 1566–1571. Read more…
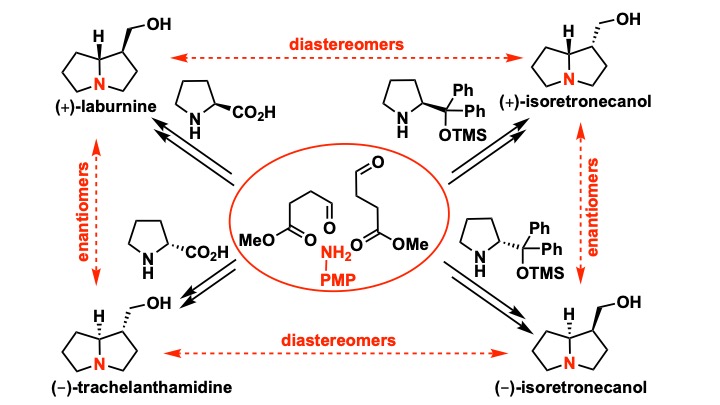
Sarkale, A. M.; Appayee, C.* “Stereodivergent Synthesis of 1-Hydroxymethylpyrrolizidine Alkaloids” Org. Lett. 2020, 22, 4355–4359. Read more…
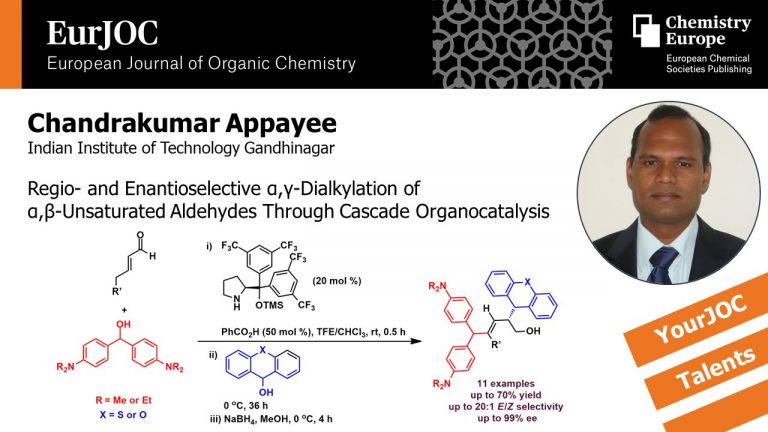
Kutwal, M. S.; Padmaja, V. M. D.; Appayee, C.* “Regio‐and Enantioselective α,γ‐Dialkylation of α,β‐Unsaturated Aldehydes through Cascade Organocatalysis” Eur. J. Org. Chem. 2020, 2720-2724. Read more…
Invited Article for a themed collection “YourJOC Talent”. Read More…
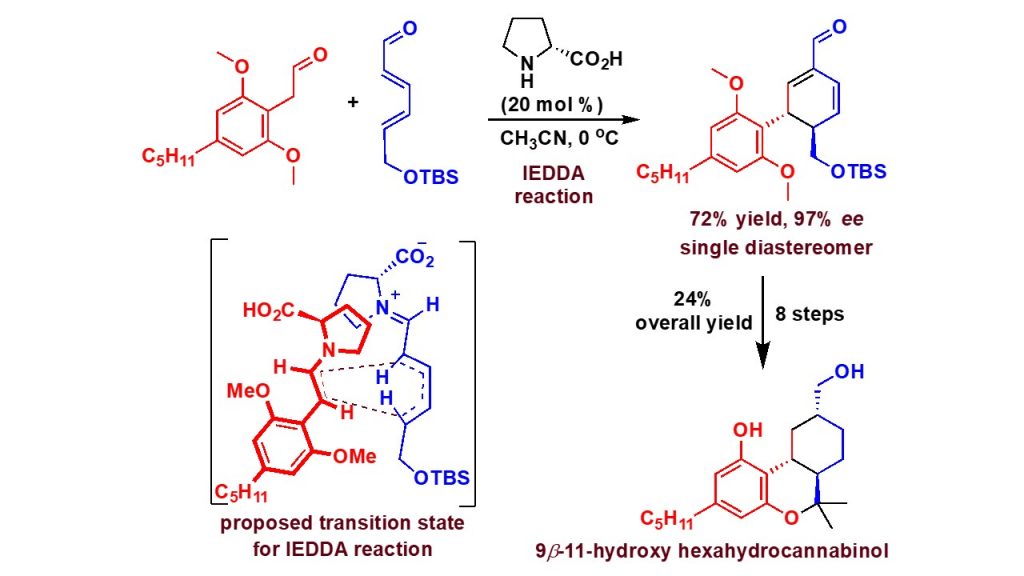
Maurya, V.; Appayee, C.* “Enantioselective Total Synthesis of Potent 9β-11Hydroxyhexahydrocannabinol” J. Org. Chem. 2020, 85,1291-1297. Read More…
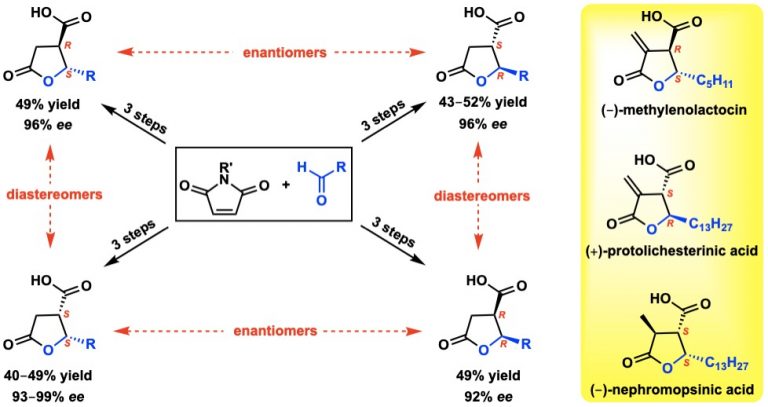
Sarkale, A. M.; Maurya, V.; Giri, S.; Appayee, C.* “Stereodivergent Synthesis of Chiral Paraconic Acids via Dynamic Kinetic Resolution of 3-Acylsuccinimides” Org. Lett. 2019, 21, 4266–4270. Read More…
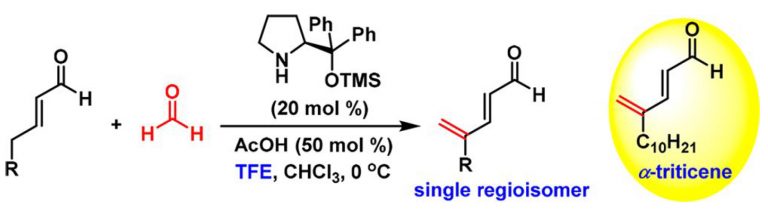
Kutwal, M. S.; Dev, S.; Appayee, C.* “Catalytic Regioselective γ-Methylenation of α,β-Unsaturated Aldehydes Using Formaldehyde via Vinylogous Aldol Condensation”Org. Lett. 2019, 21, 2509–2513. Read More…
This paper was among the most downloaded articles by the readers of Organic Letters in the month of April 2019.
This work is highlighted in Synfacts 2019, 15, 0797. Read More…
Padmaja, V. M. D.; Jangra, S.; Appayee, C.* “Highly Regioselective α-Alkylation of α,β,γ,δ-Unsaturated Aldehydes”. Org. Biomol. Chem. 2019, 17, 1714-1717. Read More…
Invited Article for a themed collection “New Talent”. Read More…
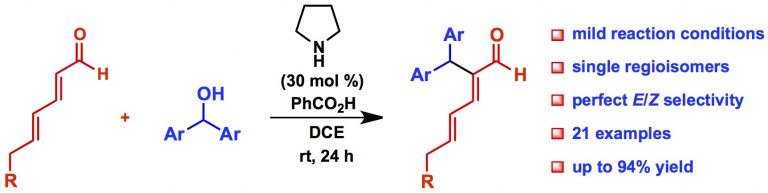
Maurya, V.; Appayee, C.* “Catalytic Asymmetric Synthesis of 3,4-Disubstituted Cyclohexadiene Carbaldehydes: Formal Total Synthesis of Cyclobakuchiols A and C”. Org. Lett. 2018, 20, 4111-4115. Read More…
This work is highlighted in Synfacts 2018, 14, 0978. Read More…
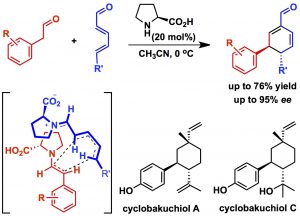
Sarkale, A. M.; Kumar, A.; Appayee, C.* “Organocatalytic Approach for Short Asymmetric Synthesis of (R)-Paraconyl Alcohol: Application to the Total Syntheses of IM-2, SCB2, and A-Factor γ-Butyrolactone Autoregulators”. J. Org. Chem. 2018, 83, 4167-4172. Read More…
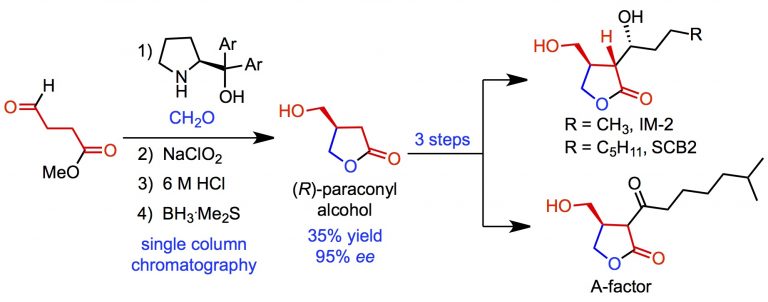
Kutwal, M. S.; Appayee, C.* “Highly Regio- and Enantioselective γ-Alkylation of Linear α,β-Unsaturated Aldehydes”. Eur. J. Org. Chem. 2017, 4230-4234. Read More…
This article is featured in the virtual issue “Emerging Investigators from India”. Read more…
This article has been highlighted on the Front cover page of the journal. Read More…
Publications from Postdoctoral and Doctoral Research
Appayee, C.; Breslow, R.* Deuterium studies reveal a new mechanism for the formose reaction involving hydride shifts. J. Am. Chem. Soc. 2014, 136, 3720–3723.
Chen, W.; Li, H.; Widowsky, J. R.; Appayee, C.; Venkataraman, L.; Breslow, R. Aromaticity decreases single-molecule junction conductance. J. Am. Chem. Soc. 2014, 136, 918–920.
Jones, J. H.; Appayee, C.; Brenner-Moyer, S. E.* One‐pot preparation of enantiopure fluorinated β‐amino acid precursors. Eur. J. Org. Chem. 2014, 5273–5280.
Breslow, R.*; Ramalingam, V.; Appayee, C. Catalysis of glyceraldehyde synthesis by primary or secondary amino acids under prebiotic conditions as a function of pH. Origins Life Evol. Biospheres 2013, 1–7.
Breslow, R*.; Appayee, C. Transketolase reaction under credible prebiotic conditions. Proc. Natl. Acad. Sci. USA 2013, 110, 4184–4187.
Appayee, C.; Fraboni, A. J.; Brenner-Moyer, S. E.* γ-Amino alcohols via organocascade reactions involving dienamine catalysis. J. Org. Chem. 2012, 77, 8828–8834.
Appayee, C.; Brenner-Moyer, S. E.* Organocatalytic enantioselective olefin aminofluorination. Org. Lett. 2010, 12, 3356–3359. (Highlighted in Synfacts, 2010, 9, 1070.)
Takenaka, N.; Chen, J.; Captain, B.; Sarangthem, R.; Chandrakumar, A. Helical chiral 2-aminopyridinium ions: A new class of hydrogen bond donor catalysts. J. Am. Chem. Soc. 2010, 132, 4536–4537. (Highlighted in Synfacts, 2010, 6, 0712.)
Prasad, K. R.*; Chandrakumar, A.; Dikundwar, A. G.; and Guru Row, T. N. Polymorphism in a TADDOL analogue induced by the presence of a chiral impurity. CrystEngComm 2010, 12, 3452–3454.
Prasad, K. R.*; Chandrakumar, A. Stereoselective synthesis of cytotoxic anhydrophytosphingosine pachastrissamine [jaspine B]. J. Org. Chem. 2007, 72, 6312–6315.
Prasad, K. R.*; Chandrakumar, A. Stereoselective syntheses of γ-alkyl (aryl)-α,β-dihydroxy-γ-butyrolactones and naturally occurring lipid guggultetrol. Tetrahedron, 2007, 63, 1798–1805.
Prasad, K. R.*; Chandrakumar, A.; Anbarasan, P. Asymmetric synthesis of both enantiomers of α-methyl-α-methoxyphenylacetic acid from L-(+)-tartaric acid: Formal enantioselective synthesis of insect pheromone (–)-frontalin. Tetrahedron: Asymmetry, 2006, 17, 1979–1984.
Prasad, K. R.*; Chandrakumar, A. Nucleophilic addition reactions of 1,4-diketones derived from tartaric acid: Synthesis of TADDOL analogues. Synthesis, 2006, 2159–2166.
Prasad, K. R.*; Chandrakumar, A. Asymmetric synthesis of α-methoxyarylacetic acid derivatives. Tetrahedron: Asymmetry, 2005, 16, 1897–1900.